You read about this all the time.
Company X buys Company Y for its new drug or medical device that has yet to receive FDA approval. The price tag is monstrous at millions or hundreds of millions of dollars.
To get payback on that investment, Company X needs an FDA approval. So, Company X launches a clinical trial to collect the critical data that will ultimately lead to its FDA approval or rejection.
It might sound like a simple process. But, running a clinical trial is a gosh darn mess. The biggest challenges with clinical trials are:
- Only 10% of clinical trials enroll on time.
- 90% of clinical trials need to DOUBLE timelines to meet enrollment targets.
- 48% of clinical trials do not meet patient recruitment goals.
- 11% of clinical sites fail to enroll one patient.
“Patient recruitment for clinical trials is a nightmare!” states a VP of Clinical Affairs at a leading medical device company. “Clinical trials are time-consuming and expensive.”
How expensive can it be to enroll JUST ONE PATIENT for a Phase III study? As reported by Moe Alsumidaie at Applied Clinical Trials, the cost to enroll a single patient can be as high as $26,000 for a Phase III study.
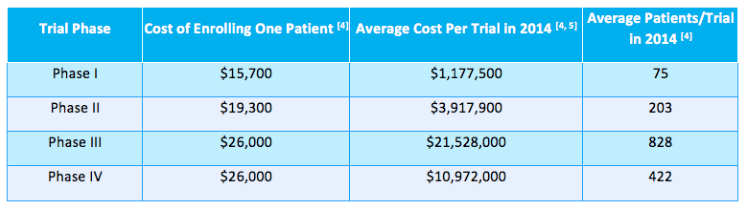 (Source: Applied Clinical Trials)
(Source: Applied Clinical Trials)
So… Patient recruitment is slow and expensive. That’s now a given. It’s also a given that for each day a new drug or device is not commercialized, millions of dollars are left on the table. Research suggests that the opportunity cost for lost drug sales can be $600K per day for niche products and over $8 MILLION PER DAY for blockbuster products.
Is $60 Million A Year About Right?
Given this difficult situation, we wanted to learn even more.
To further evaluate the expense and the lost sales for a medical device clinical trial that was enrolling 1,000 patients, we calculated that the annual costs were $60 million using analysis provided by William Maisel at TCT.

We then asked several executives to answer a very simple question, “What is the annual cost to your company to run this type of clinical trial?”
As you might imagine, we didn’t get definitive answers. So, we asked a follow-up question: “We modeled the cost to be about $60 million. How does that sound to you?”
To our surprise, the responses we received were:
- “That’s probably light.”
- “That’s conservative.”
- “More like $100 million.”
When we asked about the roadblocks to minimizing those losses and accelerating the commercialization of new products, the answers normalized quickly.
“My biggest pain point is patients,” stated a Vice President of Clinical Research at a leading medical company. “I will be running 11 clinical trials in 2019. We need a better way to find and educate patients about the benefits of clinical trials. That is the only way to speed this up. Accelerating patient recruitment is very important for me right now.”
You read about this all the time.
Company X buys Company Y for its new drug or medical device that has yet to receive FDA approval. The price tag is monstrous at millions or hundreds of millions of dollars.
To get payback on that investment, Company X needs an FDA approval. So, Company X launches a clinical trial to collect the critical data that will ultimately lead to its FDA approval or rejection.
It might sound like a simple process. But, running a clinical trial is a gosh darn mess. The biggest challenges with clinical trials are:
- Only 10% of clinical trials enroll on time.
- 90% of clinical trials need to DOUBLE timelines to meet enrollment targets.
- 48% of clinical trials do not meet patient recruitment goals.
- 11% of clinical sites fail to enroll one patient.
“Patient recruitment for clinical trials is a nightmare!” states a VP of Clinical Affairs at a leading medical device company. “Clinical trials are time-consuming and expensive.”
How expensive can it be to enroll JUST ONE PATIENT for a Phase III study? As reported by Moe Alsumidaie at Applied Clinical Trials, the cost to enroll a single patient can be as high as $26,000 for a Phase III study.
 (Source: Applied Clinical Trials)
(Source: Applied Clinical Trials)
So… Patient recruitment is slow and expensive. That’s now a given. It’s also a given that for each day a new drug or device is not commercialized, millions of dollars are left on the table. Research suggests that the opportunity cost for lost drug sales can be $600K per day for niche products and over $8 MILLION PER DAY for blockbuster products.
Is $60 Million A Year About Right?
Given this difficult situation, we wanted to learn even more.
To further evaluate the expense and the lost sales for a medical device clinical trial that was enrolling 1,000 patients, we calculated that the annual costs were $60 million using analysis provided by William Maisel at TCT.

We then asked several executives to answer a very simple question, “What is the annual cost to your company to run this type of clinical trial?”
As you might imagine, we didn’t get definitive answers. So, we asked a follow-up question: “We modeled the cost to be about $60 million. How does that sound to you?”
To our surprise, the responses we received were:
- “That’s probably light.”
- “That’s conservative.”
- “More like $100 million.”
When we asked about the roadblocks to minimizing those losses and accelerating the commercialization of new products, the answers normalized quickly.
“My biggest pain point is patients,” stated a Vice President of Clinical Research at a leading medical company. “I will be running 11 clinical trials in 2019. We need a better way to find and educate patients about the benefits of clinical trials. That is the only way to speed this up. Accelerating patient recruitment is very important for me right now.”
You read about this all the time.
Company X buys Company Y for its new drug or medical device that has yet to receive FDA approval. The price tag is monstrous at millions or hundreds of millions of dollars.
To get payback on that investment, Company X needs an FDA approval. So, Company X launches a clinical trial to collect the critical data that will ultimately lead to its FDA approval or rejection.
It might sound like a simple process. But, running a clinical trial is a gosh darn mess. The biggest challenges with clinical trials are:
- Only 10% of clinical trials enroll on time.
- 90% of clinical trials need to DOUBLE timelines to meet enrollment targets.
- 48% of clinical trials do not meet patient recruitment goals.
- 11% of clinical sites fail to enroll one patient.
“Patient recruitment for clinical trials is a nightmare!” states a VP of Clinical Affairs at a leading medical device company. “Clinical trials are time-consuming and expensive.”
How expensive can it be to enroll JUST ONE PATIENT for a Phase III study? As reported by Moe Alsumidaie at Applied Clinical Trials, the cost to enroll a single patient can be as high as $26,000 for a Phase III study.
 (Source: Applied Clinical Trials)
(Source: Applied Clinical Trials)
So… Patient recruitment is slow and expensive. That’s now a given. It’s also a given that for each day a new drug or device is not commercialized, millions of dollars are left on the table. Research suggests that the opportunity cost for lost drug sales can be $600K per day for niche products and over $8 MILLION PER DAY for blockbuster products.
Is $60 Million A Year About Right?
Given this difficult situation, we wanted to learn even more.
To further evaluate the expense and the lost sales for a medical device clinical trial that was enrolling 1,000 patients, we calculated that the annual costs were $60 million using analysis provided by William Maisel at TCT.

We then asked several executives to answer a very simple question, “What is the annual cost to your company to run this type of clinical trial?”
As you might imagine, we didn’t get definitive answers. So, we asked a follow-up question: “We modeled the cost to be about $60 million. How does that sound to you?”
To our surprise, the responses we received were:
- “That’s probably light.”
- “That’s conservative.”
- “More like $100 million.”
When we asked about the roadblocks to minimizing those losses and accelerating the commercialization of new products, the answers normalized quickly.
“My biggest pain point is patients,” stated a Vice President of Clinical Research at a leading medical company. “I will be running 11 clinical trials in 2019. We need a better way to find and educate patients about the benefits of clinical trials. That is the only way to speed this up. Accelerating patient recruitment is very important for me right now.”
You read about this all the time.
Company X buys Company Y for its new drug or medical device that has yet to receive FDA approval. The price tag is monstrous at millions or hundreds of millions of dollars.
To get payback on that investment, Company X needs an FDA approval. So, Company X launches a clinical trial to collect the critical data that will ultimately lead to its FDA approval or rejection.
It might sound like a simple process. But, running a clinical trial is a gosh darn mess. The biggest challenges with clinical trials are:
- Only 10% of clinical trials enroll on time.
- 90% of clinical trials need to DOUBLE timelines to meet enrollment targets.
- 48% of clinical trials do not meet patient recruitment goals.
- 11% of clinical sites fail to enroll one patient.
“Patient recruitment for clinical trials is a nightmare!” states a VP of Clinical Affairs at a leading medical device company. “Clinical trials are time-consuming and expensive.”
How expensive can it be to enroll JUST ONE PATIENT for a Phase III study? As reported by Moe Alsumidaie at Applied Clinical Trials, the cost to enroll a single patient can be as high as $26,000 for a Phase III study.
 (Source: Applied Clinical Trials)
(Source: Applied Clinical Trials)
So… Patient recruitment is slow and expensive. That’s now a given. It’s also a given that for each day a new drug or device is not commercialized, millions of dollars are left on the table. Research suggests that the opportunity cost for lost drug sales can be $600K per day for niche products and over $8 MILLION PER DAY for blockbuster products.
Is $60 Million A Year About Right?
Given this difficult situation, we wanted to learn even more.
To further evaluate the expense and the lost sales for a medical device clinical trial that was enrolling 1,000 patients, we calculated that the annual costs were $60 million using analysis provided by William Maisel at TCT.

We then asked several executives to answer a very simple question, “What is the annual cost to your company to run this type of clinical trial?”
As you might imagine, we didn’t get definitive answers. So, we asked a follow-up question: “We modeled the cost to be about $60 million. How does that sound to you?”
To our surprise, the responses we received were:
- “That’s probably light.”
- “That’s conservative.”
- “More like $100 million.”
When we asked about the roadblocks to minimizing those losses and accelerating the commercialization of new products, the answers normalized quickly.
“My biggest pain point is patients,” stated a Vice President of Clinical Research at a leading medical company. “I will be running 11 clinical trials in 2019. We need a better way to find and educate patients about the benefits of clinical trials. That is the only way to speed this up. Accelerating patient recruitment is very important for me right now.”


Written by Adam Pick, CEO of PatientEvolution
Adam is a patient recruitment enthusiast who has been featured in The Wall Street Journal, BusinessWeek, and Medical News Today. Adam has helped over 50 hospitals and medical device companies accelerate patient recruitment for commercial therapies and clinical trials.

Written by Adam Pick
CEO of PatientEvolution
Adam is a patient recruitment enthusiast who has been featured in The Wall Street Journal, BusinessWeek, and Medical News Today. Adam has helped over 50 hospitals and medical device companies accelerate patient recruitment for commercial therapies and clinical trials.

